What's new about Enlite: Medtronic's newest CGM sensor
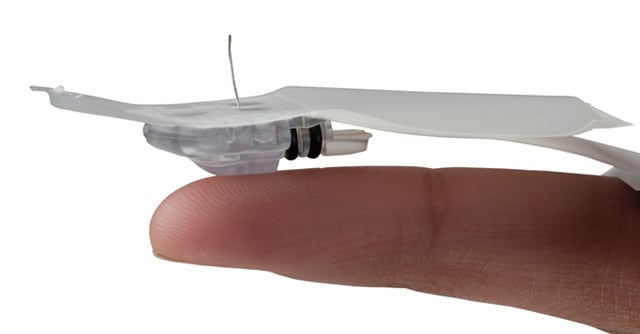
Since launching MiniMedTM 530G with Enlite, many customers have asked us how Enlite is different from previous sensors. So, we went to Jeff Myers, Product Manager from our CGM team, to give us the scoop. Here's what hehad to say.
A few years back, Medtronic initiated a research project to get a solid understanding of what you, our customers, are looking for in CGM products. We spoke with healthcare providers and customers across the nation to get a better understanding of the strengths and weaknesses of our glucose sensor, Sof-sensor. Throughout this process, we were able to uncover a group of customer needs that we, in turn, used to translate into product features for our new Enlite sensor.
These new features can be categorized in three key areas: performance, comfort and simpler insertion. Let's take a closer look at each of them:
Performance: Enlite is our most accurate sensor*[1]
With an important feature like Threshold Suspend in the MiniMedTM 530G with Enlite system, having a glucose sensor with optimal performance is key. Fortunately, with Enlite, accuracy is in its DNA! To help improve the accuracy and consistency of our glucose sensors, our research and development team made modifications to the sensor's chemistry so that the Enlite sensor would generate a stronger, more consistent sensor signal. This change also helped increase the glucose sensor's sensitivity so that it is more responsive to when your glucose changes.
Once we developed Enlite, we studied its accuracy in a randomized clinical trial. This is an important part of evaluating products before they come to market. In that trial, we saw a 31% improvement in overall accuracy compared to the previous Sof-sensor glucose sensor[2]. We also saw a sensor usage life of up to six days so patients could wear their sensor continuously, for a longer period of time.
Comfort: More comfortable, smaller and more flexible
We understand the importance of comfort when customers are wearing a glucose sensor continuously, so the Enlite sensor was designed to provide two forms of comfort. The first has to do with the sensor insertion. To help improve the comfort of inserting glucose sensors we focused on the insertion needle. Enlite sensors feature a smaller needle, approximately 10.5 mm in length, which is 38% smaller than the needle used to insert the Sof-sensor glucose sensor[3]. Additionally, the needle used to insert the Enlite sensor is a 27 gauge at its thinnest point, compared to the 22 gauge for Sof-sensor's insertion needle (when it comes to gauge, the larger the number, the smaller the needle). When we asked our clinical trial participants what they thought about how comfortable the insertion of Enlite sensor was, 84% agreed that sensor insertion for Enlite was no more painful than a fingerstick test[4].
The second part we focused on is the size of the sensor which, unlike the needle, stays inside your body. Approximately 8.5 mm in length, Enlite is 38% shorter than Sof-Sensor glucose sensors, and features 69% less insertion volume[5]. This means that the Enlite sensor is 1/3 of the size of Sof-sensor. Enlite is also more flexible, allowing the sensor to easily bend and move as your body moves, which can make it more comfortable to wear. When we asked our clinical trial participants, 80% of them agreed they did not feel the sensor underneath their skin[6].
Simpler Insertion: New simple insertion process
One of the things we've often heard from our customers is that they wanted the insertion process to be simpler. When it comes to diabetes management, simplicity is key, right? So, we made changes to improve the way you insert the sensor and the device you use for the insertion.
Enlite is inserted into your body at a 90 degree angle, the angle we've learned that most people with diabetes prefer (it's one of the reasons our Quick-set infusion set (link to Quick-set page on store) is so popular). In addition, the Enlite Serter features a new, ergonomic shape that was designed to muffle the sound when inserting a sensor on the body and hide the needle and sensor from view. You actually never see the needle during insertion! The insertion needle remains inside a spring-loaded needle housing which automatically retracts the needle when removed from the sensor base.
The Enlite Serter was so well designed it was the silver award winner in the medical device category of the IDEA program!
To get the most benefit from all these improvements, proper training is key. So as a reminder, before experiencing the Enlite, please attend an in-person training.
Have you had a chance to experience Enlite sensor? Tell us about it in the comments section below.




Blog comments
What is the ISIG range for the Enlite?
That’s a great question, Ken. I’d like to connect you with a member from my team to explain the sensor ISIG range to you so we will be in touch with you soon.
My old pump failed under warranty . It's a shame they would not let me get the new pump
Hi Bruce. I’m sorry to hear that, I will have someone from my team reach out to you to discuss any options you might have.
I would like to know how much insulin the reservoir can hold in mini-med 530G..Thanks
Hi Louis, like our Paradigm Revel system, the MiniMed 530G system comes in 2 reservoir sizes, 1.8 mL and 3 mL. Please let me know if you have any more questions!
I have been a pump + cgm user for many years and am on my 3rd or 4th pump. I do not have insurance that covers the cost of the pumps or the supplies but would be interested in upgrading to the new 530G. Would someone let me know the cost of the new pump as well as the senors and the transmitter?
Thanks
Hi Peg. Thanks for reaching out. We do offer financial assistance for those who qualify. I will have someone from my team contact you to discuss your options.
Mary, I am happy to look into this for you, and will have a member of my team connect with you. Please know we have quite a number of the New Tech Guarantee upgrades to process, so please bear with us as we work hard to provide new product and training to everyone in the program.
I signed up to "New Tech Guar Program" quiet a while ago. I have not heard from anyone to date. Iam so anxious to get the new pump 530G because i get alot of low BG while iam sleeping. I got the Revel pump Dec 2012. Can someone please help me?
Thanks. Mary
As an active/sports person and public speaker, I had the original CGM sensor loosing its signal to the pump too frequently that I stopped using it. Is there an improvement in the connection, so that, turning you torso and breaking the connection plane has been solved for in this new solution?
Steve, good question and I'm sorry to hear you had problems with our previous sensor. Our CGM team is out of the office for the holidays but let me look in to this and I’ll get back to you as soon as I can. Thanks for your patience.
Steve, the new MiniMed 530G with Enlite system includes improvements that can positively impact the connection between the transmitter and insulin pump. Keep in mind the Enlite sensor is smaller and it is important to keep the sensor secure to avoid dislodging of the sensor which could impact connectivity.
This new sensor is a complete nightmare. It wakes me up constantly throughout the night saying I'm low and turning on threshold suspend. When I test, I am not low, most of the times not even close. I calibrate it and it will still beep at me 5-10min later saying I'm low again; it never registers the updated BG I calibrated. I end up turning the sensor off half of the time I wear it, which makes me wonder why I bother. Anyone else experiencing this problem?
Hi Rachel, I am very sorry to hear that you have had a frustrating experience with the new sensor. I will have someone from my team contact you shortly.
The Enlite sensor is a vast upgrade from the soft sensor. But, the usable life of the enlite is a terrible disappointment. I have now used 8 sensors and the average life of good usable information from the sensor to the pump is only about 72 hrs. With the cost of the enlite sensors being about twice of the soft sensors and the life of the enlite being about half of the soft sensors, the 4 fold increase in user costs is very hard to live with.
Hi Dennis. I'm so sorry to hear that you have had a frustrating experience with the Enlite sensor. I will have someone from my team contact you shortly to try to help.
I will be contacting my rep, but I'm finding the same results as that of Rachel (Dec 2013 post above). After 40 years as a Type 1 & the 530G being my 3rd pump, I was ready for CGM to help level out my bGs. The Enlite is comfortable & not as inconvenient to wear as I thought it might be. However, the sensors transmit my bGs as high (200+) when my Bayer meter shows 99-115. I have been getting no sleep after 3am because my pump begins alarming for a low bG. At the first alert, I crawl out of bed to check my bG, which typically registers about 120, calibrate, & return to bed only to be reawakened with another low bG alarm. Another bG check (AND calibration) only results in another alarm, even a threshold suspend. What is the purpose of a 'calibration' if it doesn't reset the bG readings? I now have turned off threshold suspend & am wondering if CGM (this one, anyway) is even relevant to my management or my 'trends.' Note: I am staying hydrated & keeping my sensor site well away from my infusion site & I calibrate a minimum of 4x per day since the 12-hr recommendation gives completely faulty results.
By the way, SOMEone in R&D should consider engineering the backlight to come on when there is a low bG or threshold suspend alarm. This is ANOTHER source of frustration when trying to respond to the pump in the dead of night.
Cathey, I’m so sorry to hear you that you are experiencing issues with your Enlite sensor. I will have someone from my team contact you shortly to help.
I've had the same experiences as Cathay and Rachel. The CGM is best at ruining your sleep at night, not monitoring your glucose. Mine keeps telling me that my glucose is low, I'm dying and to call 911. My glucose will be 4.0mol/L, yes low but no I'm not dying. My wife wants me to sleep on the coach. I calibrate 2-3x per day. Overall the CGM product is a disappointment.
Peter, I’m sorry to hear you are frustrated with your CGM. For optimal sensor performance, we do recommend calibrating 3 – 4 times a day. I will have someone from my team contact you to see what else we might be able to do to help, too. In the meantime, you may find this helpful: http://www.medtronicdiabetes.com/customer-support/sensors-and-transmitt…. Please let me know if you have any other questions.
I have not had a full night sleep since wearing the enlite sensor due to "false" low BG alarms. Obviously many of us are having the same problem could you please post some suggestions as to what we can try?
Hi Robin. I am sorry to hear you’ve been experiencing issues with your sensor alarms. I will have a member from my team contact you to try and help. Please let me know if there’s anything else I can help you with.
I started the 530G and CGM a few days ago and have some questions/concerns with the CGM. What is the normal range for the "points" to be off? I'm finding I am always 20 or more "points" off when I check, and that could be in either direction. A minimum of a 40 "point" spread is pretty big. I will calibrate and nothing changes. I've been calibrating at least 3 times a day and when my levels are stable (per the graph). Why calibrate when the levels on the sensor don't change? I understand there is a lag time between BG and SG, but shouldn't the numbers be closer, especially when there are stable levels?
That’s a great question, Megan. When glucose levels are rising or falling quickly, there may be a larger difference between your BG and SG meter values, such as after meals or taking a bolus of insulin. You may also find this information helpful: http://www.medtronicdiabetes.com/customer-support/sensors-and-transmitt…. I will have someone from my team connect with you to try and help.
I am going to upgrade my pump soon and have came here to find user opinions. I certainly am glad I did.
I used the previous CGM system and it was terrible. I didn't like having to calibrate all the time. I was hoping to see that the new system is improved. Needless to say I am disappointed by what I've seen here so far.
I want a CGM that actually checks my sugar and does it accurately. The need for 3 to 4 times a day calibration does not make the life of a diabetic easier in which I thought was the goal. Why not make a CGM that doe what the name entails without cumbersome upkeep?
This sensor is like the old one as far as my research has concluded, only worse. Consistent interruptions in sleep for inaccurate reports of glucose levels makes it worse than its predeccaser.
Medtronic please quit grasping at straws and make a device that works as hard for the diabetic as the diabetic does to keep it correct. I have never researched another company for a insulin pump before, but I will now. My health and well being is important to me and I'll be back when I feel your products work and are not betas or experiments. I'm very disappointed because I have always like my Paradigm, but I refuse to be a Guinea Pig while Medtronic figures out how to make a true CGM.
Todd, I’m sorry to hear you are disappointed. We absolutely do care about the people who use our devices and are focused on improving the lives of people with diabetes, which is why we have invested to improve the accuracy and comfort of our sensors, and to advance insulin delivery with Threshold Suspend, an important step toward an artificial pancreas. All CGM systems require BG meter readings in order to generate sensor glucose readings. Calibrations are essential to making sure the glucose sensor maintains its accuracy over time. If I may, I’d like to suggest that you also read the guest blog, My Initial Thoughts on the MiniMed 530G with Enlite - http://www.loop-blog.com/blog/My-Initial-Thoughts-on-the-MiniMed-530G-w…. You may find this customer’s experience helpful. If I can help you in any other way please do let me know.
I would just like to start by saying that I am an Trauma Nurse in a Level One Trauma center working the night shift. I have been using the Enlite sensors now for awhile. I don't find the problems that the users above have named in regards to the alerts however, EVERY single sensor I have inserted, the needle does not retract without some type of force. I have watched the 'how - to' videos online multiple times to see if I'm doing something wrong but I'm not. I have to twist, push, pull...anything the get the needle to retract. This morning, frustrated after yet another problem with the retracting needle, I looked up online to see if anyone else was having difficulty. I found a YouTube video (http://www.youtube.com/watch?v=sDyuO7m7QV8) and this man had the EXACT same problem!!!! I point out that I am a trauma nurse for the simple fact that I insert IVs in people every hour of every shift I work, I know how to use a needle....many different types at that. There is absolutely no reason that these retractable needles should be so difficult to remove, never mind the cost of each one if you're unable to remove it all!!!! I sincerely hope this issue gets fixed....FAST!
Hi Kacy. I’m sorry to hear you’re experiencing issues with the insertion process. I’ll have one of our advanced device specialists connect with you to try and help. In the meantime, here’s a Quick Reference Guide for Using the Enlite sensor that you also may find helpful: http://www.medtronicdiabetes.com/sites/default/files/library/support/95….
Imagine hearing people talk about the poor quality of actual glucase readings the sensor supplies and then have a rep tell them that she will have someone contact them . The device is of poor quality. I do not understand how Medtronic doesn't a better quality sensor. I know a girl at work that uses a device from a different company it is always about right on her reading when she calibrates. Now you are coming out with a device that will suspend insulin when a person has a low blood sugar?
Rob, I’m sorry for any negative experiences you’ve had with the quality of the sensor as we’re really excited about the new Enlite sensor and its advancements in comfort and accuracy. Please know that the safety and durability of our products is very important to us, and we will continue to take your feedback and suggestions to implement more changes in the future. If you send me your contact information at loopblog@medtornic.com, I’d be more than happy to connect you with someone from my team to try and help.
I too am frustrated to the point of giving up. The soft-sensors while uncomfortable sometimes had far better real-life performance. I rarely get on of the new enlite sensors to last more than 3 days and even then,b the medt r onic system is more random number generator than a readout of my glucose. The sensor is always coming out bent or S-shaped. Too fragile in my opinion. I've tried new lots of sensors, new transmitters and a different pump. All without success! .
Dave, I’m sorry to hear you’re frustrated with the Enlite sensor. Since I don’t know the specifics of your case, I will have a member of my team reach out to you to try and help.
I have been using the Soft-sensor for a few years and have gotten pretty good accuracy with it. I do still have a couple of issues. One with the transmitter/sensor attachment and one with the pump alarms. First the attachment. The transmitter is way too heavy to stay attached when I sweat a lot. I've tried everything including the tackiest adhesives (Mastisol) for this available as well as using a non-scented deodorant on the site. I'm not sure if the enlite sensor/transmitter is any lighter. Secondly my largest gripe is the incessant alarming when it is telling me I need to test my glucose. Both the early warning as well as when the time comes to test it. When I am driving I cannot get to my pump and shut down the alarm due to heavy traffic and safety. I can understand this type of urgency if the alarm is for a high BG or a low BG but not for a calibration. Does the new 530G pump with the Enlite sensor have this same issue?
Thanks!
Hi Todd. I’m sorry to hear you’re having trouble with your sensor adhesion and your pump alarms. The type of adhesive for the Enlite sensor has not changed. However, the amount of tape has been increased to extend under the transmitter. It’s important to let you know that we discontinued Sof-sensors in December 2015. I will have a member of my team reach out to you help you out and discuss your options.
I love the new Enlite Sensors compared to the previous model sensors I used. Insertion is easy and quick. However, I've had a hard time recently avoiding hitting blood vessels in my abdomen. I am an average weight, but the vessels are hard to spot under the skin. 2 Enlite sensors were wasted this morning, one on each side due to bleeding. Is Medtronic working on a way to imbed a VeinViewer/AccuVein type device in the inserter? That would solve my one big issue with using the CGM!
I’m sorry to hear about your experience, Cheryl. I’ll have one of our specialists reach out to you to see if they can help. And I’m sorry but due to FDA rules I cannot discuss any potential future products.
I consider the medtronic elite sensor a disaster. The new calibration algorithm on the 630G is even worse. In the past 2 months I have had 4 of 5 sensors from one box work fine but only last 4 or 5 days, Then I had 3 of 5 sensors fail to calibrate properly at all. Actual sugar 179, sensor reading 81 with low warning. Calibrate only went up to 151 then plummeted back down to 73! Same experience on all 3. Started another box 2 nights ago, It is 630 AM no sleep for 2 nights as the sensor keeps dancing up and down low warning, calibrate high warning then low warning then calibrate again, Meantime sugar was between 148 and 171. Your sensor & calibration algorithm is not ready for release and the CDC should disqualify
I am sorry to hear you are having trouble with your sensor, Marvin. This is a good conversation to have with our 24-Hour HelpLine. Please give them a call directly at 800.646.4633, option 1, to discuss this in more detail.
Hi
Remove the sensor opstie, the sensor wire didn’t found.
I want to Know where the wire goes and how to explain this situation ?
Eric, I'm sorry to hear this. This is a conversation best addressed by our 24-Hour HelpLine. If you still need assistance, please give them a call at your convenience at 800.646.4633, option 1.
We are looking for a new CGM for my husband. This one says the needle gauge is 27 at its thinnest point. What is the gauge of the insertion needle at its thickest point?
Also, I had heard there were plans to combine the CGM with the pump itself so there would be one insertion site. Is that still happening?
Thanks for your interest, Vi. I don't have this information, but our technical support may be able to help. You can reach them at 800.646.4633, option 1. As far as new products, we're unable to discuss any information until items have been approved by the FDA. As updates are available, we'll be sure to share them with our community.
Post a new comment
Required fields are marked *