Insulin pump therapy. An advanced option for diabetes management.
Learn more about how insulin pump therapy works, the benefits of pump therapy, and who it is suitable for.
What is insulin pump therapy?
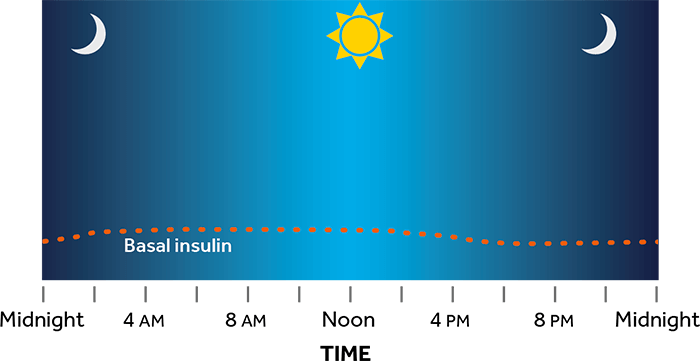
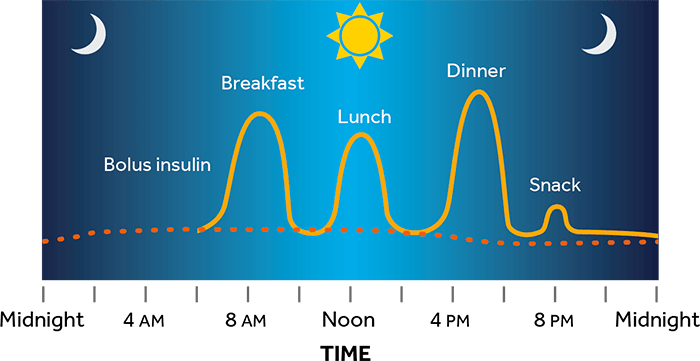
An insulin pump is a small device that can help you manage your diabetes. It delivers continuous and customized doses of rapid-acting insulin 24 hours a day to match your body’s needs. The pump provides insulin to your body in two ways:
How does an insulin pump work?
A pump delivers insulin to the body through a thin, flexible tube called an infusion set.
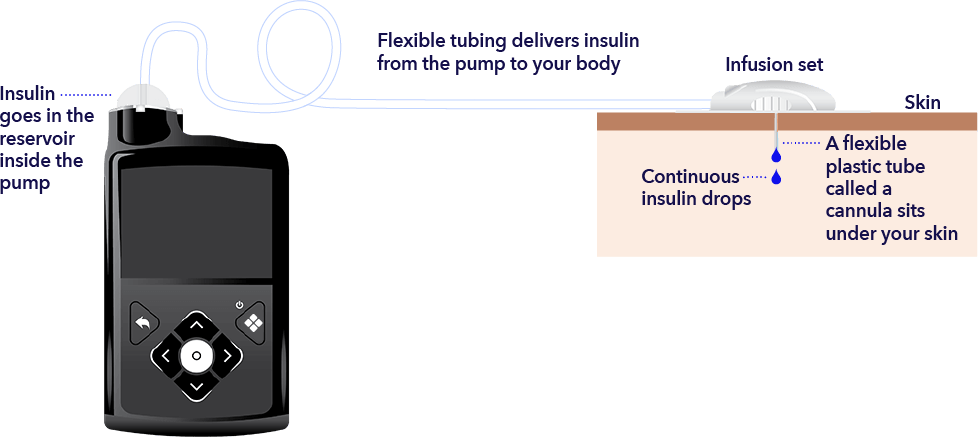
What are the benefits of insulin pump therapy?
People can experience many positive changes in their life when switching to pump therapy.
- An insulin pump holds up to 300 mL of insulin, so you don’t have to worry about carrying around all of the supplies needed for injections
- Insulin can be easily and discreetly delivered with a few button pushes
- Pumps can be programmed to deliver basal insulin at different rates throughout the day
- Change your mealtime insulin based on the food you choose to eat
- Insulin pump delivers rapid-acting insulin which minimizes insulin variability1,2
- Delivers basal and bolus insulin to more closely match each individual’s insulin needs
Who can wear an insulin pump?
Ask your healthcare professional about insulin pump therapy if you:
- Take 3 or more insulin injections per day
- Take other medications in addition to insulin to manage your diabetes
- Would like better management of your diabetes
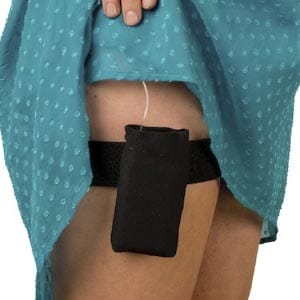
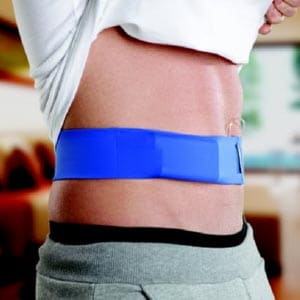
Different ways to wear an insulin pump
There are many ways a pump can be worn. It can be comfortably worn during work, exercise, formal occasions, and everyday life. Hover over the images below to see how others wear their pump.
Does insurance cover an insulin pump?
When it comes to your diabetes management, cost should not prevent you from accessing advanced diabetes technology. Our team will work with you to help ensure that you can experience the benefits of insulin pump therapy.
Private insurance‡
Most private insurance companies cover insulin pumps under the durable medical equipment portion of your policy. Depending on your insurance coverage, you might have to pay a deductible and/or percent of the cost (co-insurance). If your deductible and out-of-pocket maximum has been met, the insulin pump might be covered at 100% by your insurance.
Government insurance‡
Government insurances such as Medicare and Medicaid may cover insulin pumps depending on the state and other requirements. A patient's out-of-pocket cost under government insurance varies depending on the policy.
Insurance processing
When you start the process of getting an insulin pump, you do not have to worry about the paperwork. Medtronic will help you every step of the way by verifying your insurance, providing an estimated out-of-pocket cost, collecting the documents from you and your physician, and submitting all the required documents to your insurance company.
Payment options
If you do not have insurance or need assistance with your out-of-pocket cost, Medtronic offers flexible payment options and a financial assistance program for qualifying customers.
Frequently asked questions
MiniMed™ 780G system
The only system with Meal Detection™ technology** that provides automatic adjustments and corrections† to sugar levels every 5 minutes.§
* At the time of manufacture and when the reservoir and tubing are properly inserted, your pump is waterproof. It is protected against the effects of being underwater to a depth of up to 12 feet (3.6 meters) for up to 24 hours. This is classified as IPX8 rating. See user guide for more details. The sensor and transmitter are water-resistant at 8 feet (2.4 meters) for up to 30 minutes. CGM readings may not be transmitted from the CGM to the pump while in water.
‡ Coverage guidelines for insulin pumps vary and are subject to the insurance companies published medical policies in effect at the time services are requested.
**Taking a bolus 15 – 20 min before a meal provides significant improvement in post meal control. We recommend taking a meal bolus.
† Refers to auto correct, which provides bolus assistance. Can deliver all correction doses automatically without user interaction, feature can be turned on and off.
§ Refers to SmartGuard™ feature. Some user interaction required. Individual results may vary.
Φ Optional CGM
1. Heinemann L, Weyer C, Rauhaus M, Heinrichs S, Heise T. Variability of the metabolic effect of soluble insulin and the rapid-acting insulin anolog insulin aspart.Diabetes Care.1998;21:1910-1914.
2. Heise T, Nosek L, Ronn BB, et al. Lower within-subject variability of insulin detemir in comparison to NPH insulin and insulin glargine in people with type 1 diabetes. Diabetes. 2004;53:1614-1620.
3. Bergenstal RM,Tamborlane WV, Ahmann A, et al. Effectiveness of sensor-augmented insulin-pump therapy in type 1 diabetes. N Engl J Med. 2010;363:311–320.
4. Adults, T1 and parents of children with T1 diabetes < 18 years were surveyed.
5. Individual results may vary.
6. dQ&A US Diabetes Patient Panel Report; Customer Overall Satisfaction, n=146; Q4 2023: P.52 (November 2023).
Individuals pictured and/or quoted here were compensated for spending the day with us and allowing us to photograph them and their family. Their thoughts and opinions are their own.
Important Safety Information: MiniMed™ 780G system with SmartGuard™ technology with Instinct sensor, Simplera Sync™ sensor, and Guardian™ 4 sensor
The MiniMed™ 780G system is intended for the continuous delivery of basal insulin at selectable rates and the administration of insulin boluses at selectable rates for the management of type 1 diabetes mellitus in persons 7 years of age and older, and of type 2 diabetes mellitus in persons 18 years of age and older requiring insulin. The system is also intended to continuously monitor glucose vales in the fluid under the skin.
The MiniMed™ 780G System includes SmartGuard™ technology, which can be programmed to automatically adjust insulin delivery based on the continuous glucose monitoring (CGM) sensor glucose values and can suspend delivery of insulin when the sensor glucose (SG) value falls below or is predicted to fall below predefined threshold values. The system is intended for use with connected sensors, including the Simplera Sync™ and Guardian™ 4 sensors and integrated continuous glucose monitors, including the Instinct sensor, each of which has different wear-time, form factor, insertion site, and other distinguishing characteristics that relate to sensor performance. Consult the appropriate sensor user guide when using the system. Discuss treatment decisions with your HCP.
WARNING: Do not use the SmartGuard™ feature for people who require less than 8 units or more than 250 units of total daily insulin per day. A total daily dose of at least 8 units, but no more than 250 units, is required to operate in the SmartGuard™ feature.
WARNING: Do not use the MiniMed™ 780G system until appropriate training has been received from a healthcare professional. Training is essential to ensure the safe use of the MiniMed™ 780G system.
WARNING: Do not use SG values to make treatment decisions, including delivering a bolus, while the pump is in Manual Mode. When the SmartGuard™ feature is active and you are no longer in Manual Mode, the pump uses an SG value, when available, to calculate a bolus amount. However, if your symptoms do not match the SG value, use a blood glucose (BG) meter to confirm the SG value. Failure to confirm glucose levels when your symptoms do not match the SG value can result in the infusion of too much or too little insulin, which may cause hypoglycemia or hyperglycemia.
Pump therapy is not recommended for people whose vision or hearing does not allow for the recognition of pump signals, alerts, or alarms. The safety of the MiniMed™ 780G system has not been studied in pregnant women or in persons using other anti-hyperglycemic therapies that do not include insulin. For complete details of the system, including accessories and additional important safety information concerning indications, contraindications, warnings and precautions associated with the system and its components, please consult https://www.medtronicdiabetes.com/important-safety-information and the appropriate user guides at https://www.medtronicdiabetes.com/download-library.
 Watch video
Watch video