Medtronic announces FDA approval for MiniMed 770G insulin pump system

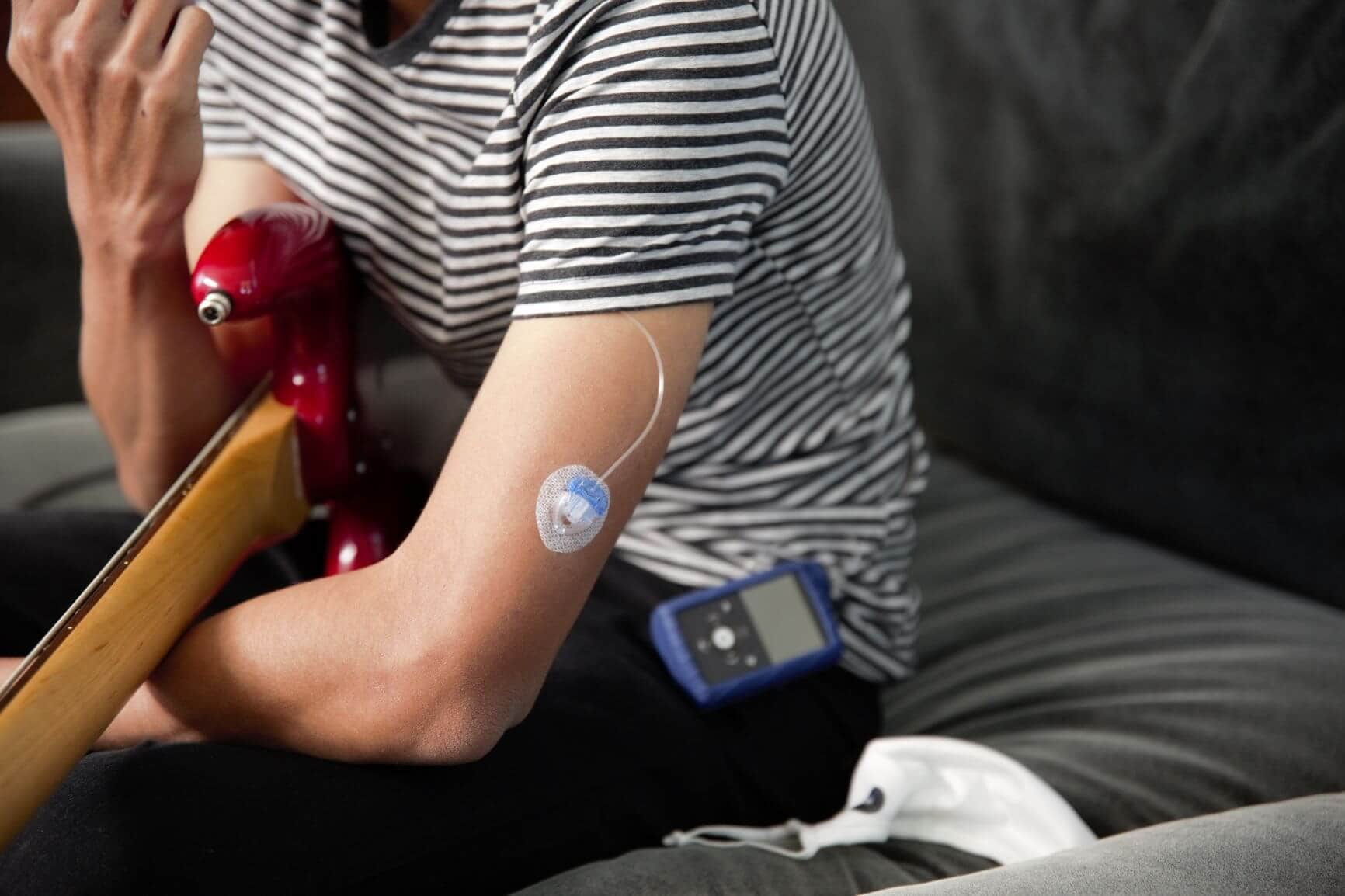
We’re excited to announce the next innovation in Medtronic Diabetes technology. On September 1st, 2020, Medtronic received FDA approval for the MiniMedTM 770G system! This new insulin pump system builds on the MiniMedTM 670G system, providing smartphone connectivity and an expanded age indication to ages 2 and up.
 With the MiniMedTM 770G system, you get the same great technology you know and love from the MiniMedTM 670G system, but now with the ability to see your sugar levels from your (or a care partner’s) smartphone. Here’s what you can expect:
With the MiniMedTM 770G system, you get the same great technology you know and love from the MiniMedTM 670G system, but now with the ability to see your sugar levels from your (or a care partner’s) smartphone. Here’s what you can expect:
- For people on the MiniMedTM 770G system, the MiniMedTM Mobile app mirrors the pump screen to allow you to see your pump and continuous glucose monitoring (CGM) data on your personal phone. You’ll also receive notifications from your system with alerts or alarms when you’re heading out of range. The MiniMedTM Mobile app works on both iPhone and Android devices.
- For care partners, the CareLinkTM Connect app provides visibility to pump and CGM data so they can view sugar levels in real time, even when not close by. The app also allows care partners to receive text notifications if sugar levels are out of range.
- Smartphone connectivity allows those living with diabetes and their care partners to view insulin delivery and bolus information on their mobile device.
- The MiniMedTM 770G system is also enabled for future software upgrades, once such software upgrades are available. Customers will be offered a software upgrade at no charge through December 2021.1
- We’re also excited to share this new system is approved for ages 2 and older!
- The MiniMedTM 770G has SmartGuardTM Auto Mode, which means it automatically adjusts2 background insulin every five minutes, 24 hours a day, based on your unique insulin needs. The goal of this automated insulin delivery is to help maximize the time glucose levels are within the target range and to help minimize highs and lows.

The MiniMedTM 770G system includes an insulin pump, a CGM system (with GuardianTM Sensor 3), diabetes management software (CareLinkTM Personal software), the MiniMedTM Mobile app, CareLinkTM Connect app, and a blood glucose meter (Roche Accu-Chek Guide Link meter). For more information on compatible devices, infusion sets, and other products, check out the technical specifications here.
Watch this video below for a message from our group president.
To learn more about the MiniMedTM 770G system and request more information, check it out here. The MiniMedTM 770G system will be available for purchase in the coming months.
1 Program eligibility requirements and terms and conditions apply. Subject to change.
2 Refers to SmartGuard Auto Mode. Some user interaction required. Individual results may vary.
Actor portrayal in these photos.
IMPORTANT SAFETY INFORMATION: MINIMED™ 770G SYSTEM WITH SMARTGUARD™ TECHNOLOGY
The MiniMed™ 770G system is intended for continuous delivery of basal insulin (at user selectable rates) and administration of insulin boluses (in user selectable amounts) for the management of type 1 diabetes mellitus in persons two years of age and older requiring insulin as well as for the continuous monitoring and trending of glucose levels in the fluid under the skin. The MiniMed™ 770G system includes SmartGuard™ technology, which can be programmed to automatically adjust delivery of basal insulin based on continuous glucose monitoring (CGM) sensor glucose (SG) values and can suspend delivery of insulin when the SG value falls below or is predicted to fall below predefined threshold values.
The Medtronic MiniMed™ 770G system consists of the following devices: MiniMed™ 770G insulin pump, the Guardian™ Link (3) transmitter, the Guardian™ Sensor (3), one-press serter, the Accu-Chek® Guide Link blood glucose meter, and the Accu-Chek® Guide test strips. The system requires a prescription.
The Guardian™ Sensor (3) has not been evaluated and is not intended to be used directly for making therapy adjustments, but rather to provide an indication of when a fingerstick may be required. All therapy adjustments should be based on measurements obtained using a blood glucose meter and not on values provided by the Guardian™ Sensor (3).
All therapy adjustments should be based on measurements obtained using the Accu-Chek® Guide Link blood glucose meter and not on values provided by the Guardian™ Sensor (3). Always check the pump display to ensure the glucose result shown agrees with the glucose results shown on the Accu-Chek® Guide Link blood glucose meter. Do not calibrate your CGM device or calculate a bolus using a blood glucose meter result taken from an Alternative Site. It is not recommended to calibrate your CGM device when sensor or blood glucose values are changing rapidly, e.g., following a meal or physical exercise.
WARNING: Do not use the SmartGuard™ Auto Mode for people who require less than 8 units or more than 250 units of total daily insulin per day. A total daily dose of at least 8 units, but no more than 250 units, is required to operate in SmartGuard™ Auto Mode. |



Blog comments
Hola!! Necesito comunicarme con ustedes por favor!!
Sure, Maria. You can email us dhelp@medtronic.com and we'll see how we can help.
No entiendo nada, no hablo ingles, pero me encantaría adquirir una bomba para mi hija, y quiero saber como o en donde buscarlas y su costo
Dalia, wed be happy to look into your options for your daughter. If you're in the US, please give our therapy specialists a call at 800.646.4633, option 3 ad we can have a Spanish-speaking teammate (or connect with an interpreter) to discuss your options. If you are outside the US, please check here http://bit.ly/1ce3pEc for local contacts who'd be happy to help.
Not everyone can have a smartphone with them every moment. So it would be helpful if your phone representatives wouldn't tell people that a smartphone is required "all the time" when asked. That would have saved me being transferred to a "lead" who seemed unhappy that I was bothering him to get corrected information. Of course, I assume this will be clearer when the manuals are actually available on-line. Not the most positive experience I've had dealing with Medtronic.
I apologize for the confusion, Howard. You will still have access to your pump screen to view your data. I'll be sure to share this feedback with our team.
I have Omnipod now but changing insurance and it will not be cost effective to stay with them so looking at options
Jonah, we appreciate the consideration. If you have any questions, please give our therapy specialists a call at 800.646.4633, option 3.
When will the MedT 670 or 770 systems (pump and CGM) be approved for coverage as DME (Durable Medical Equipment) - IE: Medicare Part B for those of us with "traditional" Medicare coverage.
Nolan, we understand how important this and are working with both the FDA and Medicare for coverage consistent with other national payors. Unfortunately, at this time, we do not have a timeline available to share, but know, we are committed to seeing this through.
Is this approved by Medicare and will Medicare cover it?
David, the new MiniMed 770G insulin pump is covered by Medicare. While our CGM is not currently covered, we do have options that can help with CGM affordability, like our CGM Access program. If you'd like to learn more, please give our therapy specialists a call at 800.646.4633, option 3.
Is it Medicare and Medicare approved. I’m currently on the 630g pump
Christopher, Medicare covers our insulin pumps and related supplies, but not our CGM products. Our CGM Access program may be able to help with your out of pocket cost. Give our therapy specialists a call at 800.646.4633, option 3 to learn more.
Will Medicare cover the 770G and CGM system? I had to swap to the XXXXXX in January 2020 because Medicare did not cover the Medtronic Guardian 3. I would love to swap back.
Carolyn, currently Medicare does not cover our CGM products, but we have programs that can help, like our CGM Access and other cost support options. If you'd like to explore your options, please give our teammates a call at 800.646.4633, option 3.
Do you offer a (Pass-Thru ) program to upgrade from my 670-G to the new 770-G?
Richard, we do offer several in-warranty upgrade options for our customers. Eligibility depends on several factors, but our therapy specialists would be happy to look into your options. Please give our team a call at 800.646.4633, option 3.
Will this mean those with the 670 G get an automatic upgrade?
John, our upgrades are not automatic, but if you're interested in exploring your in-warranty upgrade options, please give our teammates a call at 800.646.4633, option 3.
Is there going to be a upgrade program from 670G to 770G? What about a software update to convert the 670G so it can be viewed on the smartphone
Bill, upgrade options are available for our customers who are currently in-warranty, subject to eligibility. You can learn more by calling and speaking with a therapy specialist at 800.646.4633, option 3. Unfortunately, our MiniMed 670G system does not have the necessary internal components for software updates or Bluetooth connectivity.
Is there any difference in how it works and how the 570 works besides that it can be used with a smartphone?
I would really love a pump that combines manual mode with auto mode.
Patty, thanks for reaching out. The MiniMed 770G system uses the same algorithm and core technology as the MiniMed 670G system. In addition to smartphone connectivity for individuals and their care partners, it also includes over the air upgradable software and an expanded age indication for ages 2 and up.
Will you have a new holster for the 770G? When I moved up to the 670G they do not have a holster that fits 5he new pump. I use the holster that clips to your pants.
Brenda, at this time, there is no information regarding an available holster. We've shared similar requests with our development team and will be sure to share updates with our community as they become available.
Brenda, I use a flip phone case I found at Ocean State Job Lot.
Do you still have to calibrate?
Jim, yes, 3-4 calibration per day will still be required.
I've had the 670g for a couple years now. What's the process like if possible upgrading to the 770g?
Richard, that would depend on what upgrade programs you may be eligible for. To look into your options, please give our therapy specialists a call at 800.646.4633, option 3.
I really believe this machine would really help me
We'd be happy to look into your options, Michelle. Please give our therapy specialists a call at 800.646.4633, option 3 to learn more.
Hello I have a 670G pump is it compatible or it has to be the 770G?
Photina, I'm not sure I understand the question. Which compatibility are you looking into?
Hello, i've had the 670G for 3 months ((have used the 530 and 630 for 6 years) and am working with a product specialist weekly.. I do need to speak to someone in R/D dept. Many improvements can be made to the software to make the unit better streamline. Do you have the contact info?
We appreciate you reaching out, AL. Our research and development team does not have an inbound call queue, so there isn't a number we can provide. However, we'd love to hear your thoughts. Please send them over by email to dhelp@medtronic.com and we'll make sure they are shared.
The improvement you need to work on is the reliability of the Guardian Sensors so that Medicare will pay for them and realize they are capable for dosing.
Jan, we are working with the FDA on updated labeling and will share updates as they become available.
Hello,
If I’m using a Mini Med 670G would my insurance cover the cost if I went to this new pump. I’m on Medicare with Aetna as my supplemental insurance. Thanks
Rick, we'd be happy to look into your coverage and explore your options with you. Please give our therapy specialists a call at 800.646.4633, option 3 to learn more.
What brands, makes, models of smartphones are or will be supported with this system?
Great question, Bb. We are getting the final details together and will share a full list of compatible devices as we get closer to our commercial launch.
I simply want to know if Medicare will pay for this new pump. Thank you.
Mary, the new MiniMed 770G insulin pump is covered by Medicare. While our CGM is not currently covered by Medicare, please call us to discuss your options at 800.646.4633, option 3.
Does Medicare cover this yet? In relation to the CGM?
Not yet, Christina, but we are working on it.
Within a mo. of being dx with Type 1 diabetes, I began using your pumps. Been 22 yrs now and I love Medtronic! My Endo put me on a CGM that I hate. It malfunctions constantly and my primary dr and my Endo have tried to get me a Medtronic CGM but my Medicare/UHC ins. refuses to cover it. I’m on a 630G pump now and am interested in ur new 770G that comes with a CGM. Won’t u PLEASE try to help me with this. I don’t want to wear a CGM that says my bG is 49 and a finger stick with Medtronic glucometer says it’s 172. That’s dangerous! I constantly have to calibrate plus replace sensors a few times a month. Do I have to go back to 15 finger sticks a day? Thanks very much.
Jane, we'd be happy to look into your options. Please know, Medicare does not currently cover our CGM products, but our cost support options may be able to help. Please give us a call at 800.646.4633, option 3 to discuss your options.
Love the system and have been very pleased. But, I want to comment on two design items I would love for Metronic to consider.
1. The sound of pressing the buttons on the pump are too loud. In the middle of the night, or in a setting where it's quiet, the "snapping of the buttons" are very distracting. I always wake my wife up if I have to tend to the pump in the middle of the night. Can those buttons not be more quiet in operation?
2. The "violent" nature of the vibration is amazing. I think the pump vibrates way to harshly and although I think it's because Medtronic doesn't want any "accidents" to happen because the patient was unaware, I would like a softer vibration. It's jarring as it is.
Otherwise, I love the CGM and it has improved my A1C's a lot. Thanks for hearing my concerns.
Pagination
Post a new comment
Required fields are marked *